Plastic Surgery &
Non-Surgical Blog
Blog Categories
Have Questions?
We’re here to help! Read our blogs or call the office today!
Breast
Body
Face
Healthy Living
Latest Blogs
Life Changing Liposuction at Greer Plastic Surgery
In the ever-evolving landscape of cosmetic surgery, liposuction stands as a transformative procedure that continues to captivate the attention of those seeking to sculpt and redefine their bodies. In plastic surgery, we’re always...

When Is Surgery a Better Option Than Non-Surgical Treatments?
Most people need some combination of non-surgical and surgical treatments to achieve optimal results and reach their aesthetic goals. For example, fillers and laser are great preventative treatments that still play an important...
Dr. Greer’s Signature Mastopexy Technique
A breast lift can provide you with enhanced confidence, and scaling up with implants can be a highly desirable option. With Dr. Jennifer Greer’s signature mastopexy technique, which combines the benefits of both an augmentation and a...

Should I Get BOTOX® Before My Wedding?
What Beauty Treatments Should I Get Before My Wedding? BOTOX® Fillers Laser treatments Kybella or CoolSculpting Facial and dermaplaning What Can Bridal BOTOX® Do for You? While BOTOX® and other neurotoxins are a great way to treat...

Should You Have an En Bloc Capsulectomy?
If you’re asking this question, you are most likely researching the different methods for breast implant removal, a common occurrence for those who’ve had implants. Types of implant removal methods include simply removing the implant,...

Monsplasty and How to Get Rid of the Fupa
Fupa stands for fat upper pubic area (okay, so there’s a more r-rated version of the acronym, but this is what we’re sticking with). It’s often confused with a pannus, which is that apron-like overhang of skin on the lower abdomen. But...

Your Complete Guide to Lip Filler
Introduction to lip fillers Lip fillers are one of the most popular procedures I do in the office. According to the American Society of Plastic Surgeons, 2.6 million filler treatments were performed in 2020. If you’ve ever wondered if lip...
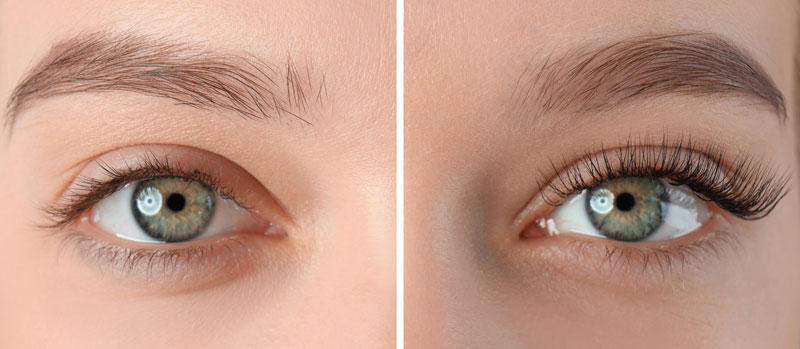
Lash Growth Serums
When Latisse entered the market in 2008, women were no longer beholden to mascara or lash extensions to have long, lush lashes. But how does Latisse work? And what are the side effects? Also, what about the other lash serums on the...

How Do I Know If I Need a Breast Reduction
Breast reduction surgery involves removal of skin, fat, and glandular tissue to make the breasts smaller and perkier. It has a very high patient satisfaction rate: 95% of patients would have the surgery again. You may be a good candidate...

Do Fillers Hurt?
Dermal fillers are one of the most popular ways to look younger without surgery. One question I hear a lot is: do fillers hurt? Although there is a little discomfort, getting fillers injected is much less painful than you may think! Your...
Greer Plastic Surgery
6101 Heisley Road
Mentor, Ohio 44060
(440) 336-8373